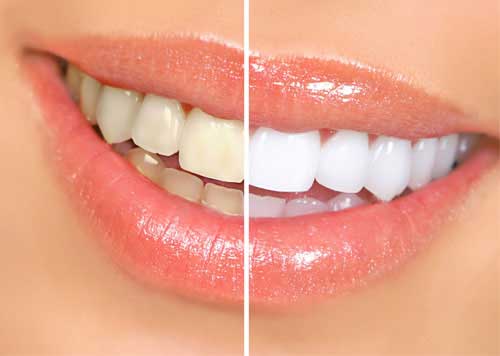
TEETH WHITENING
Tooth whitening has become one of the most frequently requested dental procedures by the public. The public has come to demand whiter, more perfect smiles and in response many choices for tooth whitening have been made available. These include home-based products such as toothpastes, gels, and films, as well as in-office based systems where products containing highly concentrated bleaching agents are applied under professional supervision. The profession and public have been aware of certain risks related to tooth whitening such as increased tooth sensitivity and gingival irritation. New research has shown that there are other risks such as tooth surface roughening and softening, increased potential for demineralization, degradation of dental restorations, and unacceptable color change of dental restorations. The new research is also focused on optimizing whitening procedures to reduce tooth sensitivity and to increase the persistence of the whitening.
Whitening chemistry
Tooth whitening is any process that lightens the color of a tooth. Whitening may be accomplished by physical removal of the stain or a chemical reaction to lighten the tooth color. Bleaching is defined here as the chemical degradation of the chromogens. The active ingredient in most whitening products is hydrogen peroxide (H2O2) which is delivered as hydrogen peroxide or carbamide peroxide. Carbamide peroxide is a stable complex that breaks down in contact with water to release hydrogen peroxide. Because carbamide peroxide releases hydrogen peroxide the chemistry of most tooth whitening is that of hydrogen peroxide.
Tooth stains consist of compounds that have color or darker shades called chromogens that are accumulated in the tooth (intrinsic) or on the tooth (extrinsic). Chromogens fall into two categories: large organic compounds that have conjugated double bonds in their chemical structure and metal containing compounds. Bleaching of the organic compounds by hydrogen peroxide involves reacting with the double bonds to oxidize the double bond. This causes the chromogen to become a lighter colored compound. Bleaching of the metallic compounds is much more difficult; better esthetic options may be veneers, bonding, or crowns. There are some professional products that contain sodium hypochlorite (NaOCl) which reacts with the double bonds of the chromogen in much the same way as peroxide
Types of Teeth Whitening Systems
Whitening systems can be variously categorized. The following approach is in accordance with the American Academy of Cosmetic Dentistry.
Whitening Toothpastes
Whitening toothpastes typically contain higher amounts of abrasives and detergents than standard toothpastes, to remove tougher stains. Whitening toothpastes do not contain bleach (sodium hypochlorite) but some contain low concentrations of carbamide peroxide or hydrogen peroxide that help lighten tooth color. Whitening toothpastes typically can lighten tooth color by about one or two shades.
OTC Whitening Strips and Gels
Whitening strips were introduced into the market in the late 1980s. They deliver a thin layer of peroxide gel on plastic strips shaped to fit onto the buccal surfaces of the teeth. There are a variety of white strip products on the market with varying instructions. A typical set of instructions are to apply the strips twice daily for 30 minutes for 14 days. Tooth lightening can be seen in several days and this method can lighten the teeth by 1 or 2 shades. There are some newer whitening strip products that require only one 30-minute application per day that have the same whitening end point as the two a day products.
Whitening gels are peroxide-based gels applied with a small brush directly to the surface of the teeth. Manufacturers instructions are usually twice a day applications for 14 days. Like the whitening strips, the teeth can usually be lightened by 1 or 2 shades.
Whitening Rinses
Whitening rinses contain oxygen sources such as hydrogen peroxide to react with the chromogens. Manufacturers instructions are for twice a day rinsing for 60 seconds each. It takes up to 3 months to see a 1 or 2 shade improvement in tooth color.
Tray-Based Tooth Whiteners
Tray-based tooth whitening systems are available both professionally and OTC. This method involves use of a fitted tray containing carbamide peroxide-bleaching gel worn for 2 to 4 hours a day or overnight. Usually by following the manufacturer instructions tooth whitening is noticeable in a few days, lightening the teeth by 1 or 2 shades.